Our "No Return Necessary"
Money-Back Guarantee
If you don’t like something of ours, guess what happens next?
No, we don’t request you deliver it to a PO box in the Gobi Desert by carrier pigeon. Nor do we ask you to fill a cursed inkwell with orc’s blood and demon saliva and then use it to complete reams of return forms written in ancient Cyrillic script.
We just . . . wait for it . . . give you your money back. Holy moo cows. And that means you can say "yes" now and decide later.
Will Genesis revolutionize every aspect of your health and wellness in 30 days flat? No.
Will it turbocharge your metabolism, hormones, and immune system? Absolutely not.
But will Genesis provide you with adaptogens, herbs, and other phytonutrients that boost energy levels and wellbeing and reduce symptoms of stress and fatigue? Yes. Or your money back.
- Total formulation transparency (no proprietary blends)[8]
- Made in the USA with globally sourced ingredients in NSF-certified and FDA-inspected and cGMP-compliant facilities
- Backed by our “No Return Necessary” money-back guarantee that works like this: If you don’t absolutely love Genesis, just let us know, and we’ll give you a full refund on the spot. No forms or returns necessary.
So order now, try Genesis risk free, and see for yourself why it’s one of the most popular all-natural greens supplements in the world (over 65,000 bottles sold and counting!).
Will Genesis revolutionize every aspect of your health and wellness in 30 days flat? No.
Will it turbocharge your metabolism, hormones, and immune system? Absolutely not.
But is Genesis the only[1] 100% natural[2] greens supplement with clinically effective doses[3] of five scientifically proven ingredients[4] for better energy levels, mood, libido, cardiovascular health, and immunity?
And will it support the health of vital organs, enhance physical performance, and reduce symptoms of stress and fatigue?
Yes. Or your money back.
- 57 peer-reviewed scientific studies support Genesis’s combination of ingredients and doses[5]
- Contains no artificial sweeteners, flavors, food dyes, or other chemical junk[6]
- Analyzed for purity and potency in a state-of-the-art ISO 17025 accredited lab[7]
- Total formulation transparency (no proprietary blends)[8]
- Made in the USA with globally sourced ingredients in NSF-certified and FDA-inspected and cGMP-compliant facilities
Genesis is also backed by our “No Return Necessary” money-back guarantee that works like this:
If you don’t absolutely love Genesis, just let us know, and we’ll give you a full refund on the spot. No forms or returns necessary.
So order now, try Genesis risk free, and see for yourself why we believe it’s the perfect all-natural greens supplement (that has sold over 65,000 bottles and counting!).
Notice to California Consumers
WARNING: Consuming this product can expose you to chemicals including lead which is known to the State of California to cause cancer and birth defects or other reproductive harm. For more information go to www.P65Warnings.ca.gov/food.
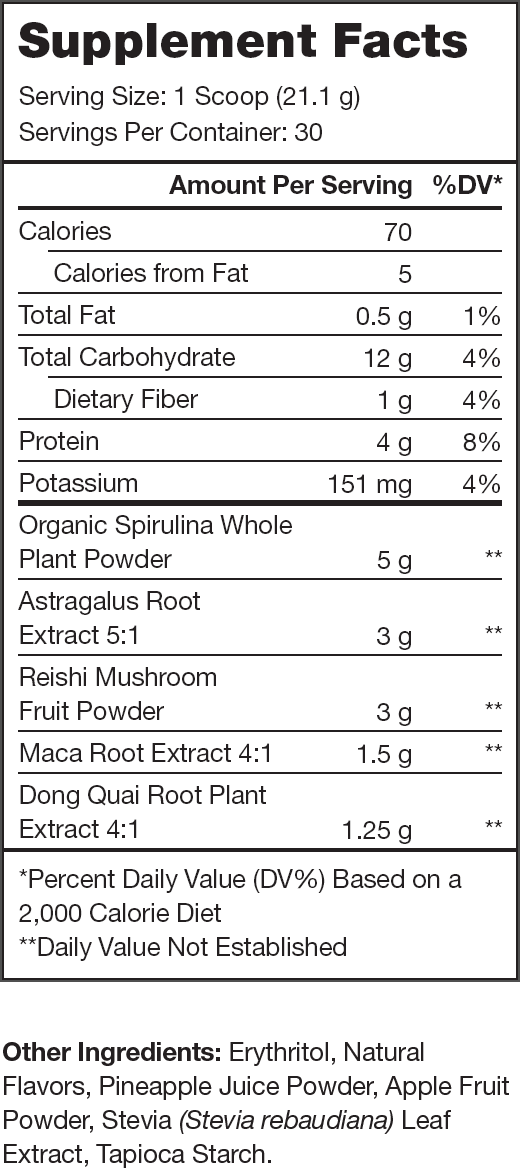
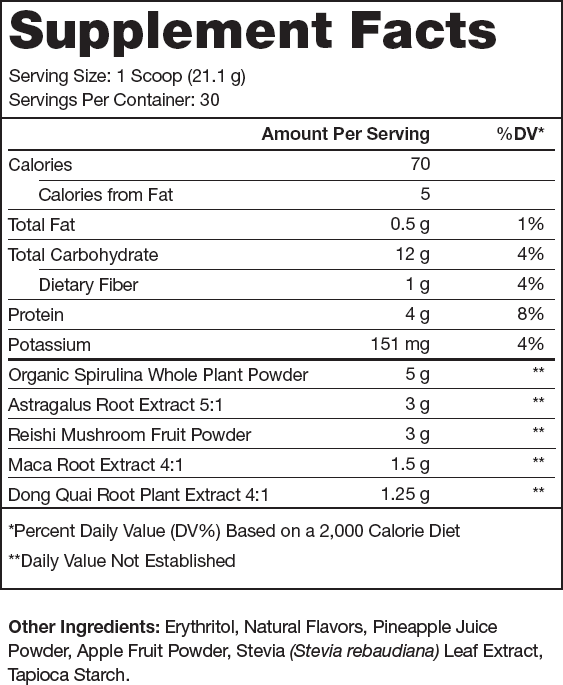
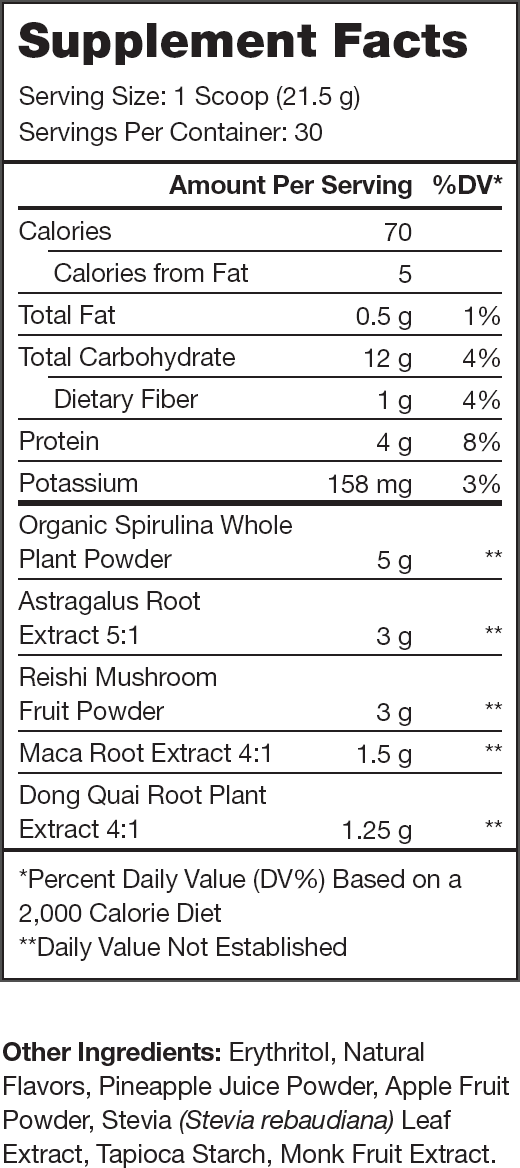
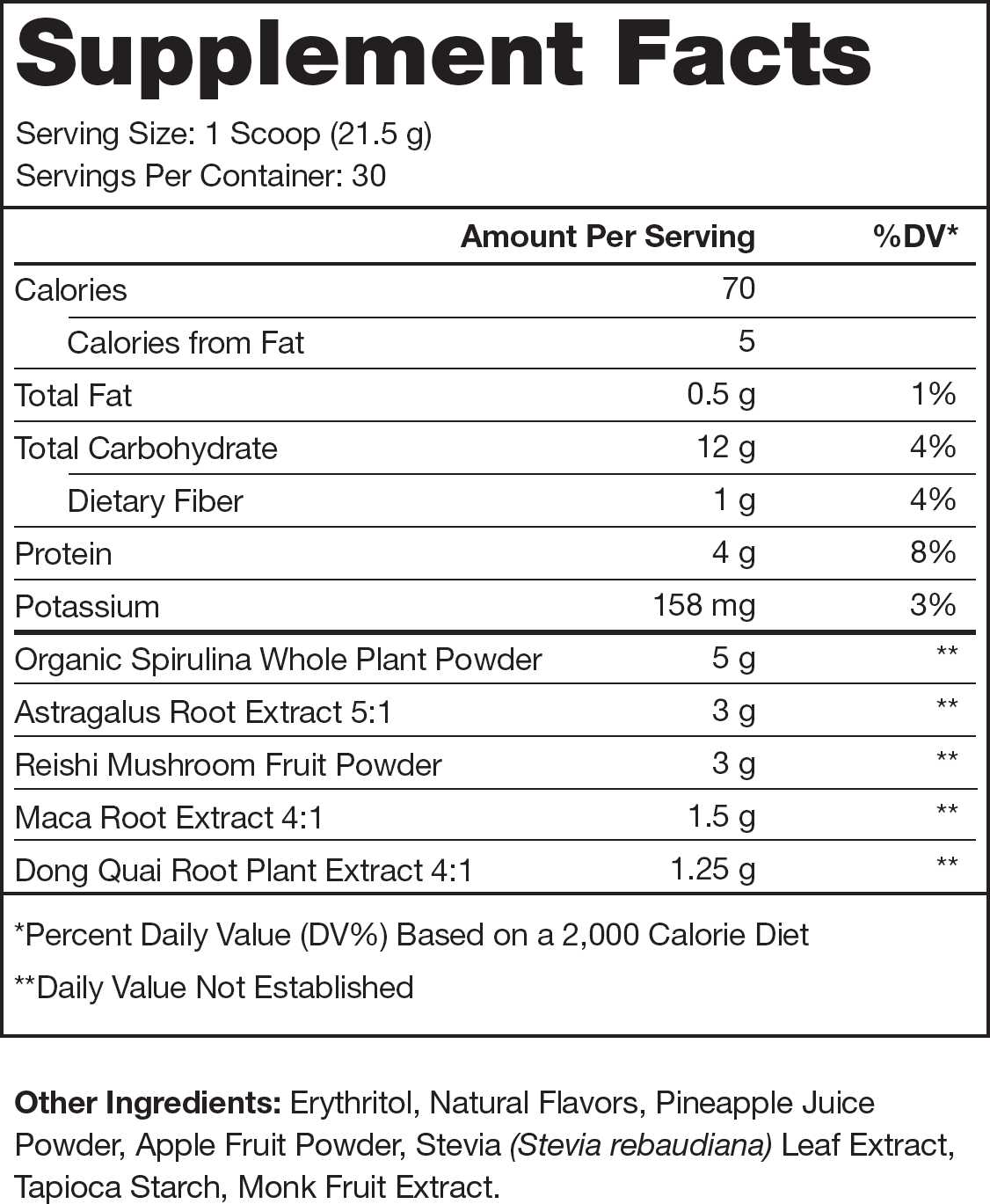
Ingredients (13.75 grams per serving)
Reishi Mushroom (3 grams per serving)
Reishi mushroom (also known as lingzhi mushroom) is a mushroom that has been used in traditional Chinese medicine for at least 2,000 years to treat a variety of conditions, including insulin resistance, immune deficiencies, fatigue, and cancer.[9]
It contains a number of molecules that have a variety of positive effects in the body, and that’s why research shows that supplementation with reishi mushroom . . .
- Helps protect liver health[10]
- Protects DNA from oxidative damage, which is a major factor in aging[11]
- Inhibits the creation of new fat cells[12]
- Reduces the time it takes to fall asleep[13]
- Helps protect brain health[14][15]
- Improves blood glucose control[16]
- Raises “good” (HDL) cholesterol levels[17]
- Boosts and balances the immune system[18][19]
- Helps protect kidney health[20]
- Has powerful anti-cancer effects[21][22]
The clinically effective dose of reishi mushroom extract is between 1.5 and 5 grams.

Spirulina (5 grams per serving)
Spirulina is a blue-green algae that’s one of nature’s richest and most complete sources of vital nutrients.
It’s often used as a vegan source of protein and is particularly abundant in B vitamins, minerals, and essential fatty acids, as well as a powerful antioxidant and anti-inflammatory molecule known as phycocyanin.
Research shows that supplementation with spirulina . . .
- Improves the cholesterol profile[23]
- Increases muscle endurance[24]
- Increases strength[25]
- Alleviates and even eliminates nasal symptoms caused by allergies[26]
- Reduces muscle damage caused by exercise[27]
- Helps the body eliminate heavy metals[28]
- Lowers blood pressure[29]
- Helps protect liver health[30]
- Reduces systemic inflammation[31]
- Improves insulin sensitivity[32]
- Enhances immunity[33]
The clinically effective dose of spirulina is between 2 and 10 grams, with most benefits seen in the range of 5 to 10 grams.

Astragalus Membranaceus (3 grams per serving)
Astragalus membranaceus (also known as Mongolian milkvetch) is a herb that has long been used in traditional Chinese medicine to increase stamina, vitality, and longevity, and to treat the cold and flu.
It contains a variety of beneficial molecules such as flavanoids and polysaccharides, but one of the more notable components is known as astragaloside IV, which helps protect cells against oxidative stress.[34]
Research shows that supplementation with Astragalus membranaceus . . .
There’s also preliminary evidence suggesting that Astragalus membranaceus may promote longevity by improving the structural integrity of DNA.[40][41]
The clinically effective dose of Astragalus membranaceus is 15 grams of the raw root, and Genesis contains 3 grams of a 5:1 Astragalus membranaceus extract per serving, providing the equivalent of 15 grams of the raw root.

Angelica Sinensis (1.25 grams per serving)
Angelica sinensis, also known as Dong Quai, is a herb popular in Traditional Chinese Medicine and commonly paired with Astragalus membranaceus for supporting vitality and organ health.
Research shows that supplementation with Angelica sinensis . . .
- Protects kidney health[42]
- Enhances blood flow[43][44]
- May protect blood vessels from inflammatory damage[45]
- May reduce systemic inflammation[46]
The clinically effective dose of Angelica sinensis isn’t established yet, but research suggests that 3 to 5 grams of the raw plant is sufficient. Additionally, when taken alongside Astragalus membranaceus, Angelica sinensis is required at a ratio of at least 1:5.
Genesis contains 1.25 grams of a 4:1 Angelica sinensis extract per serving, providing the equivalent of 5 grams of the raw plant.
Maca (1.5 grams per serving)
Maca is a plant native to Peru that has been cultivated for thousands of years for its root, which was an integral part of the diet and commerce of the ancient Incan civilization.
It contains several types of molecules known as alkaloids, which cause a number of positive effects in the body and are responsible for maca’s beneficial effects.
Research shows that supplementation with maca . . .
- Improves subjective sense of well-being[47]
- Improves sexual function in men and women[48]
- Improves sperm production and health[49]
- Improves libido in men and women[50][51]
- Can reduce feelings of anxiety and nonclinical depression[52]
The clinically effective dose of maca extract is between 1 and 3 grams.

100% Naturally Sweetened & Flavored
While artificial sweeteners may not be as dangerous as some people claim, studies suggest that regular consumption of these chemicals may indeed be harmful to our health.[53][54][55][56][57][58]
That’s why we use the natural sweeteners stevia and erythritol instead. Studies show that these ingredients are not only safe but can also confer several health benefits, including better insulin sensitivity, enhanced cholesterol profile, lower inflammation levels, and more.[59][60][61][62]

No Artificial Food Dyes or Other Chemical Junk
Studies show that artificial food dyes may cause negative effects in some people, including gastrointestinal toxicity and behavioral disorders.[63][64][65][66][67]

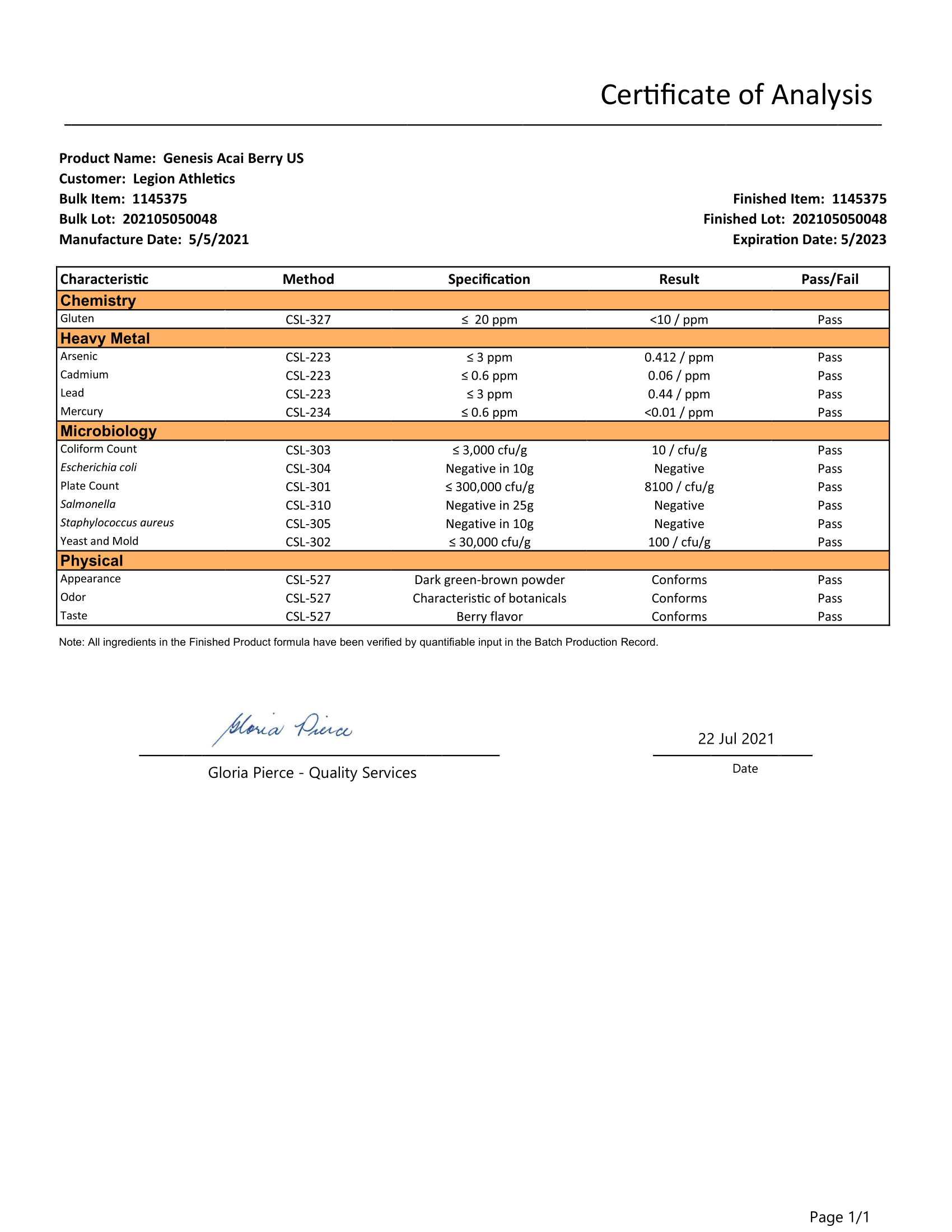
Lab Tested for Potency & Purity
Every bottle of Genesis is analyzed in a state-of-the-art ISO 17025 accredited lab to verify what is and isn’t in it. That way, you know exactly what you’re getting and putting into your body.
See how Legion compares to the rest.
- Active Ingredients
- Clinically Effective Ingredients & Doses
- Reishi Mushroom
- Spirulina
- Astragalus Membranaceus
- Angelica Sinensis
- Maca
- Natural Ingredients
- Naturally Sweetened & Flavored
- Lab Tested
- Labdoor Certified Brand
- Price Per Serving
-
Legion
Genesis
- 13.75 g
per serving - 3 g
per serving - 5 g
per serving - 3 g
per serving - 1.25 g
per serving - 1.5 g
per serving - $
-
Athletic
Greens
- 10.31 g
per serving
(proprietary blend)
(proprietary blend)
(proprietary blend)- $3.30
-
Organifi
Greens Juice
- 7.05 g
per serving
(proprietary blend)- $2.33
-
1stPhorm
Opti-Greens 50
- 9.03 g
per serving
(proprietary blend)- $2.17
The #1 brand of all-natural sports supplements.
Over 4,000,000 bottles sold to over 800,000 customers who have left us over 45,000 5-star reviews.
No Chemical Junk
Genesis doesn’t just “contain natural ingredients”—every ingredient is naturally sourced. We don’t use artificial or synthetic substances of any kind.
Clinically Effective Ingredients & Doses
Every ingredient and dose (important!) in Genesis is backed by peer-reviewed scientific research demonstrating clear benefits.
Naturally Sweetened & Flavored
Genesis is naturally sweetened and flavored with healthy, plant-based sweeteners and flavors.
Lab Tested
Genesis is tested by third-party labs for heavy metals, microbes, allergens, and other contaminants to ensure it meets FDA purity standards.
Made in USA with Globally Sourced Ingredients
Genesis is proudly made in America in NSF-certified and FDA-inspected facilities in accordance with the Current Good Manufacturing Practice (cGMP) regulations.
"No Return Necessary"
Money-Back Guarantee
If you don't absolutely love Genesis, you get a prompt and courteous refund. No forms or returns necessary.
Trusted by scientists, doctors, and everyday fitness folk alike.
Ingredients & Nutrition
- Original
- Acai Berry
Suggested Use
Mix 1 serving with 10-12 oz of cold water, or your favorite beverage. Use first thing in the morning as a great way to jump start your day!
Warning
Consult your physician prior to consumption if you have any pre-existing medical conditions, or if you are taking any prescription medication(s). Not recommended for individuals taking anti-coagulant or blood pressure medicines.
KEEP OUT OF REACH OF CHILDREN. STORE IN A COOL, DRY PLACE. DO NOT USE IF SAFETY SEAL IS BROKEN OR MISSING.
Frequently Asked Questions
+References
Some popular greens supplements are all-natural. Some contain the right mix of high-quality ingredients. Some provide clinically effective doses. But only Genesis checks each of these boxes. ↑
Genesis doesn’t just “contain natural ingredients''—every ingredient is naturally sourced from plants and animals. Genesis contains no artificial or synthetic substances of any kind.↑
Every serving of Genesis contains 13.75 grams of active ingredients that have been shown to be safe and effective in peer-reviewed scientific research.↑
Each active ingredient in Genesis is backed by published scientific studies that show benefits in healthy humans.↑
That’s 525 pages of scientific research that shows Genesis works exactly like we say it does.↑
While these types of chemicals may not be as dangerous as some people claim, studies suggest that regular consumption of them may indeed be harmful to our health. And that’s why you won’t find them in Genesis.↑
Every bottle of Genesis is guaranteed to provide exactly what the label claims and nothing else—no heavy metals, microbes, allergens, or other contaminants.↑
This means you know exactly what’s in every serving of Genesis—every dose of every ingredient—and can verify the accuracy and efficacy of the formulation.↑
That’s 525 pages of scientific research that shows Genesis works exactly like we say it does.↑
While these types of chemicals may not be as dangerous as some people claim, studies suggest that regular consumption of them may indeed be harmful to our health. And that’s why you won’t find them in Genesis.↑
Every bottle of Genesis is guaranteed to provide exactly what the label claims and nothing else—no heavy metals, microbes, allergens, or other contaminants.↑
This means you know exactly what’s in every serving of Genesis—every dose of every ingredient—and can verify the accuracy and efficacy of the formulation.↑
Bhagwan S. Sanodiya, Gulab S. Thakur, Rakesh K. Baghel, Godavarthi B. K. S. Prasad, and Prakash S. Bisen, Current Pharmaceutical Biotechnology 10, no. 8 (2009): 717–42. doi: 10.2174/138920109789978757#sthash.o6gZXyOO.dpuf. ↑
Yanling Shia, Jie Sunb, Hui He, Hui Guo, and Sheng Zhang, Journal of Ethnopharmacology 117, no. 3 (2008): 415–19. doi: 10.1016/j.jep.2008.02.023. ↑
Sissi Wachtel-Galor, Siu-Wai Choi, and Iris F. F. Benzie, Redox Report 10, no. 3 (2005): 145–49. doi: 10.1179/135100005X57355. ↑
Anita Thyagarajan-Sahu, Brandon Lane, and Daniel Sliva, Complementary and Alternative Medicine 11 (2011): 74. doi:10.1186/1472-6882-11-74. ↑
Kazuki Honda, Y. Komoda, and Shojiro Inoué, Reports of the Institute for Medical and Dental Engineering 22 (1988): 77–82. ↑
Cora Sau-Wan Lai, Man-Shan Yu, Wai-Hung Yuen, Kwok-Fai So, Sze-Yong Zee, and Raymond Chuen-Chung Chang, Brain Research 1190 (2008): 215–24. doi: 10.1016/j.brainres.2007.10.103. ↑
Ding Hui, Zhou Ming, Zhang Rui-Ping, and Xu Sheng-Li, Acta Physiologica Sinica 62, no. 6 (2010): 547–54. ↑
Yihuai Gao, Jin Lan, Xihu Dai, Jingxian Ye, and Shufeng Zhou, International Journal of Medicinal Mushrooms 6, no. 1 (2004): doi: 10.1615/IntJMedMushr.v6.i1.30. ↑
Tanya T. W. Chu, Iris F. F. Benzie, Christopher W. K. Lam, Benny S. P. Fok, Kenneth K. C. Lee, and Brian Tomlinson, British Journal of Nutrition 107, no. 7 (2012): 1017–27. doi: 10.1017/S0007114511003795. ↑
Ying Zhang, Z. Lin, Y. Hu, and F. Wang, British Journal of Sports Medicine 42, no. 10 (2007): 819–22. ↑
Lieuwe G. van der Hem, J. Adam van der Vliet, C. Frans M. Bocken, Kohsuke Kino, Andries J. Hoitsma, and Wil J. M. Tax, Transplantation 60, no. 5 (1995): 438–43. ↑
Narisa Futrakul, Tasanee Panichakul, Punnee Butthep, Prasit Futrakul, Pim Jetanalin, Suthiluk Patumraj, and Prasong Siriviriyakul, Clinical Hemorheology and Microcirculation 31, no. 4 (2004): 267–72. ↑
Shiro Oka, Shinji Tanaka, Shigeto Yoshida, Toru Hiyama, Yoshitaka Ueno, and Masanori Ito, Hiroshima Journal of Medical Sciences 59, no. 1 (2010): 1–6. ↑
Yihuai Gao, Shufeng Zhou, Wenqi Jiang, Min Huang, and Xihu Dai, Immunological Investigations 32, no. 3 (2003): 201–15. doi: 10.1081/IMM-120022979. ↑
Hee Jung Park, Yun Jung Lee, Han Kyoung Ryu, Mi Hyun Kim, Hye Won Chung, and Wha Young Kim, Annals of Nutrition and Metabolism 52 (2008): 322–28. doi: I:10.1159/000151486. ↑
Maria Kalafati, Athanasios Z. Jamurtas, Michalis G. Nikolaidis, Vassilis Paschalis, Anastasios Theodorou, Giorgios Sakellariou, Yiannis Koutedakis, and Dimitris Kouretas, Medicine & Science in Sports & Exercise 42, no. 1 (2010): 142–51. doi: 10.1249/MSS.0b013e3181ac7a45. ↑
Chaouachi M, Gautier S, Carnot Y, et al. J Diet Suppl. (2020): 1–16. doi:10.1080/19390211.2020.1832639. ↑
Cemal Cingi, Meltem Conk-Dalay, Hamdi Cakli, and Cengiz Bal, European Archives of Oto-Rhino-Laryngology 265, no. 10 (2008): 1219–23. doi: 10.1007/s00405-008-0642-8. ↑
Hsueh-Kuan Lu, Chin-Cheng Hsieh, Jen-Jung Hsu, Yuh-Kuan Yang, and Hong-Nong Chou, European Journal of Applied Physiology 98, no. 2 (2006): 220–26. ↑
Mir Misbahuddin, A. Z. M. Maidul Islam, Salamat Khandker, Ifthaker-Al-Mahmud, Nazrul Islam, and Anjumanara, Clinical Toxicology 44, no. 2 (2006): 135–41. doi: 10.1080/15563650500514400. ↑
Patricia V. Torres-Duran, Aldo Ferreira-Hermosillo, and Marco A. Juarez-Oropeza, Lipids in Health and Disease 6, no. 33 (2007): doi: 10.1186/1476-511X-6-33. ↑
E. M. Gorban, M. A. Orynchak, N. G. Virstiuk, L. P. Kuprash, T. M. Panteleĭmonova, and L. B. Sharabura, Likars’ka Sprava 6 (2000): 89–93. ↑
Panam Parikh, Uliyar Mani, and Uma Iyer, Journal of Medicinal Food 4, no. 4 (2001): 193–99. doi:10.1089/10966200152744463. ↑
Nielsen, C. H., Balachandran, P., Christensen, O., Pugh, N. D., Tamta, H., Sufka, K. J., Wu, X., Walsted, A., Schjørring-Thyssen, M., Enevold, C., & Pasco, D. S. (2010). Planta medica, 76(16), 1802–1808. https://doi.org/10.1055/s-0030-1250043. ↑
Ren S, Zhang H, Mu Y, Sun M, Liu P. J Tradit Chinese Med = Chung i tsa chih ying wen pan. 2013;33(3):413-416. http://www.ncbi.nlm.nih.gov/pubmed/24024343. ↑
Heather Zwickey, Julie Brush, Carolyn M. Iacullo, Erin Connelly, William L. Gregory, Amala Soumyanath, and Randal Buresh, Phytotherapy Research 21, no. 11 (2007): 1109–12. doi: 10.1002/ptr.2207. ↑
Bao-Mei Shao, Wen Xua, Hui Dai, Pengfei Tu, Zhongjun Li, and Xiao-Ming Gao, Biochemical and Biophysical Research Communications 320, no. 4 (2004): 1103–11. doi:10.1016/j.bbrc.2004.06.065. ↑
Qing-you Yang, Shu Lu, and Hui-ru Sun, Chinese Journal of Integrative Medicine 17, no. 2 (2011): 146–49. doi: 10.1007/s11655-011-0647-9. ↑
Deqing Wang, Yuan Zhuang, Yaping Tian, Graham Neil Thomas, Mingzhong Ying, and Brian Tomlinson, Oxidative Medicine and Cellular Longevity 2012 (2012): doi: 10.1155/2012/282383. ↑
Mingxin Li, Weixin Wang, Jun Xue, Yong Gu, and Shanyan Lin, Journal of Ethnopharmacology 133, no. 2 (2011): 412–19. doi: 10.1016/j.jep.2010.10.012. ↑
de Jesus BB, Schneeberger K, Vera E, Tejera A, Harley CB, Blasco MA. Aging Cell. 2011;10(4):604-621. doi:10.1111/j.1474-9726.2011.00700.x. ↑
Salvador L, Singaravelu G, Harley CB, Flom P, Suram A, Raffaele JM. Rejuvenation Res. 2016;19(6):478-484. doi:10.1089/rej.2015.1793. ↑
Zhang DW, Wang SL, Wang PL, et al. J Ethnopharmacol. 2020;248. doi:10.1016/j.jep.2019.112354. ↑
Dong WG, Liu SP, Zhu HH, Luo HS, Yu JP. World J Gastroenterol. 2004;10(4):606-609. doi:10.3748/wjg.v10.i4.606. ↑
Fung FY, Wong WH, Ang SK, et al. Phytomedicine. 2017;32:88-96. doi:10.1016/j.phymed.2017.04.004. ↑
Dong WG, Liu SP, Zhu HH, Luo HS, Yu JP. World J Gastroenterol. 2004;10(4):606-609. doi:10.3748/wjg.v10.i4.606. ↑
Zhuang SR, Chiu HF, Chen SL, et al. Br J Nutr. 2012;107(5):712-718. doi:10.1017/S000711451100345X. ↑
Teo Zenico, Arrigo F. G. Cicero, Linda Valmorri, Melissa Mercuriali, and Eduard Bercovich, Andrologia 41, no. 2 (2009): 95–99. doi: 10.1111/j.1439-0272.2008.00892.x. ↑
Ibid.; Nicole A. Brooks, Gisela Wilcox, Karen Z. Walker, John F. Ashton, Marc B. Cox, and Lily Stojanovska, Menopause 15, no. 6 (2008): 1157–62. ↑
Gustavo F. Gonzales, Amanda Cordova, Carla Gonzales, Arturo Chung, Karla Vega, and Arturo Villena, Asian Journal of Andrology 3, no. 4 (2001): 301–03. ↑
Gustavo F. Gonzales, Amanda Córdova, Karla Vega, Arturo Chung, Arturo Villena, Carmen Góñez, and Sonia Castillo, Andrologia 34, no. 6 (2002): 367–72. doi: 10.1046/j.1439-0272.2002.00519.x. ↑
Christina M. Dording, Lauren Fisher, George Papakostas, Amy Farabaugh, Shamsah Sonawalla, Maurizio Fava, and David Mischoulon, CNS Neuroscience & Therapeutics 14, no. 3 (2008): 182–91. doi: 10.1111/j.1755-5949.2008.00052.x. ↑
Brooks NA, et al., Beneficial Effects of Lepidium meyenii (Maca), 1157-62. ↑
Basson AR, Rodriguez-Palacios A, Cominelli F. Front Nutr. 2021;8:746247. Published 2021 Sep 24. doi:10.3389/fnut.2021.746247.↑
Suez J, Korem T, Zilberman-Schapira G, Segal E, Elinav E. Gut Microbes. 2015;6(2):149-155. doi:10.1080/19490976.2015.1017700.↑
Qin X. Department of Surgery, University of Medicine and Dentistry of New Jersey, New Jersey Medical School, Newark, New Jersey, USA. Can J Gastroenterol. 2011 Sep;25(9):511. ↑
Shil A, Chichger H. Int J Mol Sci. 2021;22(10):5228. Published 2021 May 15. doi:10.3390/ijms22105228.↑
Suez J, Korem T, Zeevi D, et al. Nature. 2014;514(7521):181-186. doi:10.1038/nature13793.↑
Frankenfeld CL, Sikaroodi M, Lamb E, Shoemaker S, Gillevet PM. Ann Epidemiol. 2015;25(10):736-42.e4. doi:10.1016/j.annepidem.2015.06.083.↑
Yadav SK, Guleria P. CSIR-Institute of Himalayan Bioresource Technology, Palampur, 176061, HP, India. Crit Rev Food Sci Nutr. 2012;52(11):988-98. ↑
Shivanna N, Naika M, Khanum F, Kaul VK. Department of Applied Nutrition, Defence Food Research Laboratory, Mysore, India. J Diabetes Complications. 2013 Mar-Apr;27(2):103-13. ↑
World Health Organization. WHO Press; 2006. Available at: http://whqlibdoc.who.int/publications/2006/9241660546_eng.pdf. Accessed January 24, 2019. ↑
Ozbayer C, Kurt H, Kalender S, Ozden H, Gunes HV, Basaran A, Cakmak EA, Civi K, Kalender Y, Degirmenci I. Department of Medical Biology, Faculty of Medicine, Eskisehir Osmangazi University, Eskisehir, Turkey. J Med Food. 2011 Oct;14(10):1215-22. ↑
Feng J, Cerniglia CE, Chen H. Division of Microbiology, National Center for Toxicological Research, US Food and Drug Administration, AR , USA. Front Biosci (Elite Ed). 2012 Jan 1;4:568-86. ↑
Kanarek RB. Department of Psychology, Tufts University, Medford, Massachusetts, USA. Nutr Rev. 2011 Jul;69(7):385-91. ↑
Nigg JT, Lewis K, Edinger T, Falk M. Oregon Health and Science University, Portland, OR, USA. J Am Acad Child Adolesc Psychiatry. 2012 Jan;51(1):86-97.e8. ↑
McCann D, Barrett A, Cooper A, Crumpler D, Dalen L, Grimshaw K, Kitchin E, Lok K, Porteous L, Prince E, Sonuga-Barke E, Warner JO, Stevenson J. School of Psychology, Department of Child Health, University of Southampton, Southampton, UK. Lancet. 2007 Nov 3;370(9598):1560-7. ↑
Gao Y, Li C, Shen J, Yin H, An X, Jin H. Scientific and Technological College of Chemistry and Biology, Yantai Univ., Yantai, PR China. J Food Sci. 2011 Aug;76(6):T125-9. ↑